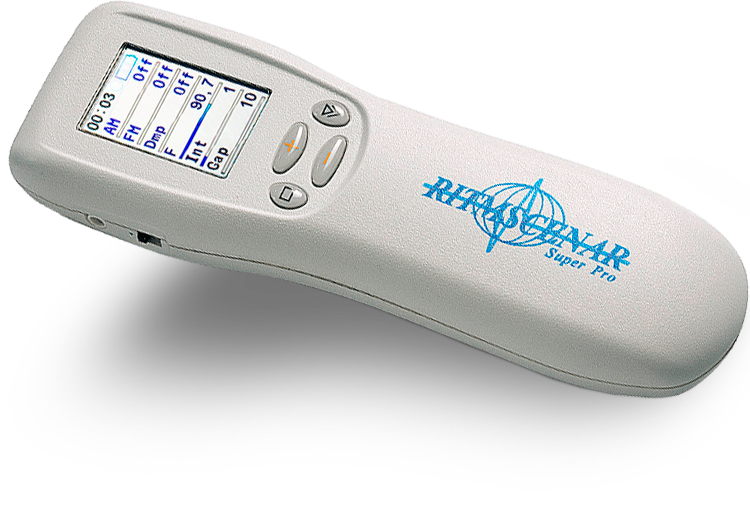
30 years ago a group of Russian scientists, doctors, engineers and physicians from Special Construction Bureau “RITM”, began developing the SCENAR or the Self-Controlled-Energo-Neuro-Adaptive Regulator invented by A. Karasev and A. Revenko. Their goal was to develop an energy efficient, multi-applicable, portable, non-invasive regulator of bodily function, perhaps even suitable to accompany cosmonauts into space.
RITM OKB is the inventor and patent holder of the famous SCENAR technology – derived from participation in the Russian National Program of space research during the 80’s
SCENAR technology as a therapy was born in 1978. The method of treatment was invented byYury Gorfinkle, Alexander Revenko and many others. The SCENAR device is capable of recognizing, treating and preventing many health problems. Russians pioneers were the first to successfully achieve repeatable therapeutic results using electrical signals to stimulate the human immune and nervous systems, using modulations of its own endogenous energy.
In 1986, the first SCENAR device, having passed technical and clinical trials, was permitted by USSR Medical Council for application in the clinic-policlinic institutions and at homes.
“SCENAR” is a trademark (according to class – therapeutic device and apparatus, electro-stimulators), registered in the state list of trademarks in USSR from 23 October 1989 by RITM.
Further lab research has been conducted in Taganrog, Ekaterinburg and Chelyabinsk. In the last 12 years, more than 20 modifications have been developed ranging from the simplest hand-held devices to the most complex computer applications. SCENAR and its creators have won the highest state awards in medical science.
SCENAR become available to the international medical community after the Soviet Union collapse and has since been gaining wide popularity and application. Thousands of medical and health practitioners in the UK, United States, Canada, Europe, and Australia now use Scenar devices.